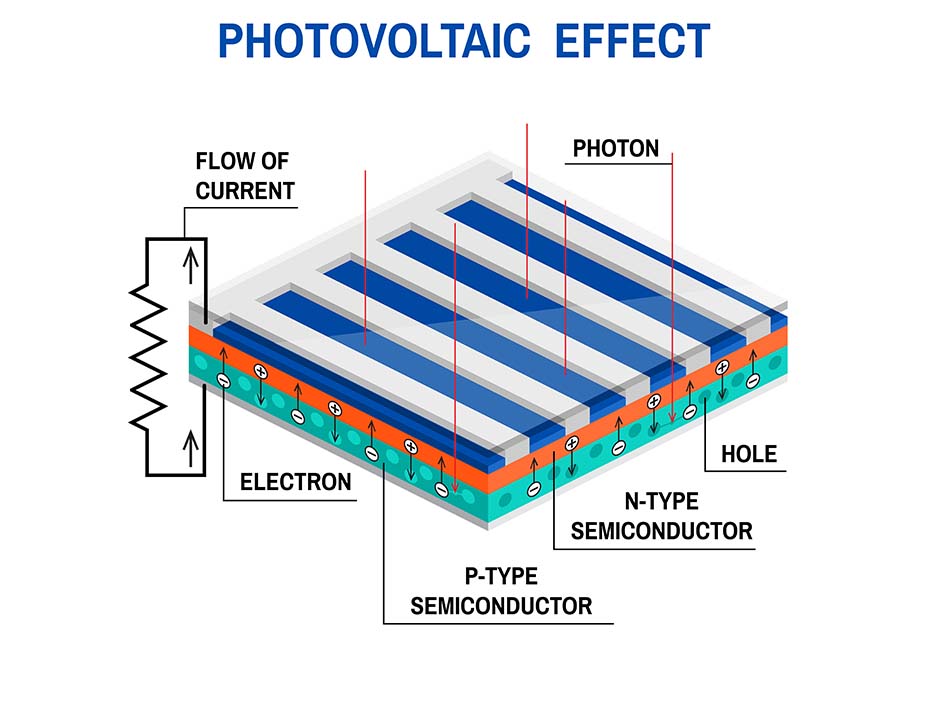
Solar (photovoltaics) cells are small electrical devices that can convert the energy from solar radiation into usable electrical energy. You can think of them as modules, which a solar panel on your roof consists of. They are connected in a specific way to obtain desirable electrical parameters of the photovoltaics panel, such as voltage, current and power. They produce direct current (DC), which later can be used to power various DC devices such as LED lighting and mobile phone batteries, or can be converted to alternate current (AC) by solar inverters and used in households.
Solar cells are most commonly made with silicon which is doped with various elements such as selenium or germanium to increase their efficiency. Depending on the silicon structure we can divide solar cells into three main types:
- mono-crystalline
- poly-crystalline
- amorphous
In mono-crystalline cells, silicon atoms create just one uniform crystalline structure. They are the most efficient solar cells on the present market, but also the most expensive ones. They are the most common types of solar cells used in the energy industry.
Poly-crystalline solar cells consist of many different smaller silicon crystals which are connected to each other. Their efficiency is slightly lower than in the mono-crystalline cells.
Amorphous cells are the least efficient, although the cheapest solar cells out of the most commonly used. The silicon within the amorphous cells does not form crystalline structures.